Texas Jury Awards $61M in Products Liability Case Against Honda After Car Wreck |
Home - Olympus Scope Lawsuit
The Carlson Law Firm is helping patients nationwide who have been diagnosed with an infection after undergoing a procedure with an Olympus Scope. Schedule a free consultation with our Olympus Scope Lawsuit lawyers at 800-359-5690.
Olympus scopes and related endoscope accessories have been linked to infection concerns, FDA safety communications, import alerts, adverse-event reporting failures, and lawsuits involving serious post-procedure infections. The FDA has warned that improper or incomplete reprocessing of certain Olympus endoscope accessories can expose patients to contaminated devices, which may result in infection, urinary tract infection, sepsis or death. Our Olympus Scope Lawsuit lawyers can help you get justice if you were seriously infected by one of these medical devices.
Patients who developed a serious infection after a procedure involving an Olympus scope may have legal options.
Free consultations and no out-of-pocket costs with our legal team.
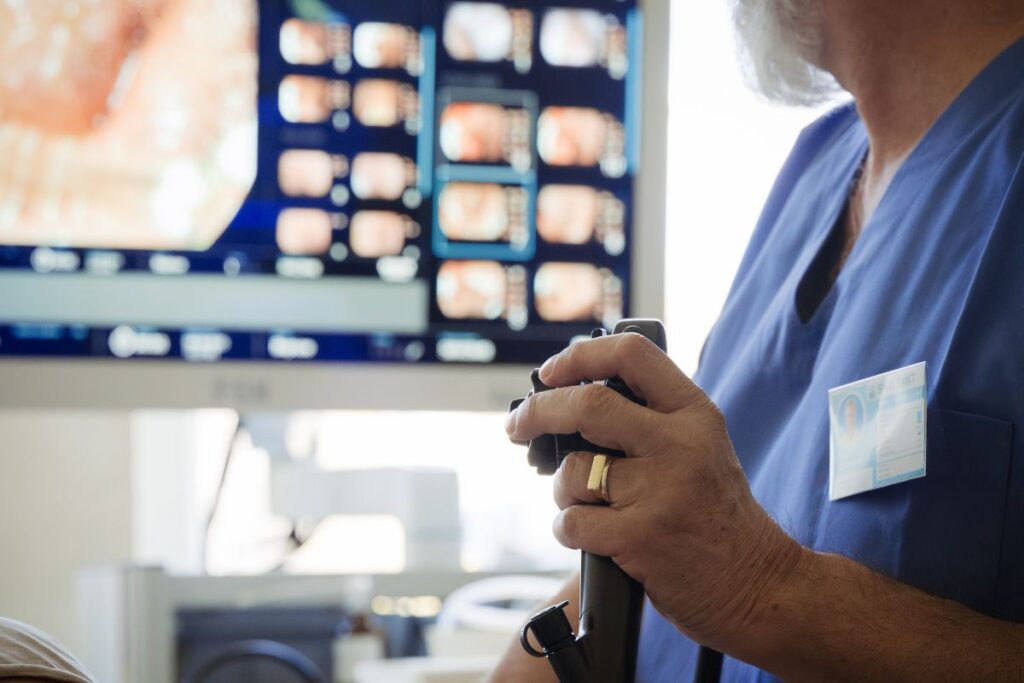
Patients who underwent procedures involving Olympus endoscopes may be eligible to pursue a claim if they experienced complications linked to contaminated or defective devices. These cases often center on infections that developed after routine diagnostic or therapeutic procedures, particularly when there is reason to believe the scope was not properly sterilized or was inherently difficult to clean due to its design.
You may qualify for an Olympus scope lawsuit if:
In some cases, family members of individuals who passed away due to complications linked to these infections may also have grounds to pursue a wrongful death claim.
Because Olympus scopes have been the subject of multiple safety alerts and investigations, eligibility does not always depend on proving improper use by a healthcare provider. Some claims focus on whether the device’s design made it unreasonably difficult to disinfect, thereby increasing the risk of cross-contamination between patients.
If you are unsure whether your experience may be connected to an Olympus scope, let us review your medical records to clarify whether you may have a claim.
Olympus manufactures medical scopes and accessories used in procedures involving the digestive tract, lungs, urinary system and other areas of the body. These devices are often reused on multiple patients after they are cleaned, disinfected and sterilized.
The concern is that certain Olympus scopes or accessories may be difficult to fully clean. If bacteria, tissue, fluid or other biological material remains inside the device, the next patient may be exposed to dangerous contamination.
Potential lawsuits may allege that Olympus failed to:
In 2018, the U.S. Department of Justice announced that Olympus Medical Systems Corporation and a former senior executive pleaded guilty to failing to file required adverse event reports involving infections linked to Olympus duodenoscopes. The DOJ said the unreported events included patient infections involving Pseudomonas aeruginosa and E. coli at medical centers in Europe.
Olympus Scopes are reusable high-definition medical cameras that can be inserted into the body to diagnose and treat disease. Patients may have come into contact with them during procedures that required endoscopes, bronchoscopes, or duodenoscopes.
Examples of when Olympus Scopes may be used:
If you underwent any of the above procedures and were diagnosed with an infection afterward, schedule a free consultation with a member of the Olympus Scopes Lawsuit team. Consultations are free, and there are no out-of-pocket costs to pursue justice.
Olympus Scopes have a design that makes it extremely difficult, or in some cases impossible, to fully sterilize. Biological material can remain trapped in crevices even after proper cleaning. Bacteria can still survive even when hospitals follow the manufacturer’s disinfection protocols. The remaining bacteria create a situation where several patients can be exposed to a scope that carries a dangerous bacterium.
After undergoing procedures for unrelated conditions, a patient may later present with an infection. Often, the infection is not immediately connected to the endoscopic procedure. Hospitals continue to unknowingly use these reusable devices, and infections continue to spread to additional patients.
Olympus manufacturers control a large share of the endoscope market. As such, there is a significant risk for widespread outbreaks. A further complication is that these scopes often involve drug-resistant organisms, which may worsen infection outcomes.
Our team has extensive experience handling claims where a product or medication caused a serious infection. Schedule your free consultation with our Olympus Scope Lawsuit lawyers. There are no out-of-pocket costs associated with a filing a lawsuit with our law firm.
Federal litigation against Roblox is moving forward as courts, state officials and families question whether the platform has done enough to protect children from sexual abuse and exploitation.
Patients may have a potential claim if they developed a serious infection or complication after a procedure involving an Olympus scope or Olympus endoscope accessory.
Reported injuries may include:
Not every infection after a medical procedure means a device was defective or contaminated. However, serious infections that occur shortly after a scoped procedure may require a closer look at the device used, the cleaning process and whether the hospital received any manufacturer or FDA warnings.
Olympus scopes and endoscope accessories have been the subject of FDA warnings involving infection risks, reprocessing concerns and quality system violations. These warnings may be important for patients who developed serious infections after procedures involving Olympus devices.
One current concern involves the Olympus MAJ-891 Forceps/Irrigation Plug, an accessory used with certain endoscopes. The FDA warned that infections may occur when the device is not disconnected from the endoscope and disassembled before reprocessing. Exposure to a contaminated device may lead to infection, urinary tract infection, sepsis and, in some cases, death.
Olympus notified U.S. customers about the issue in December 2024 after complaints involving patient injuries and one death.
In June 2025, the FDA alerted health care providers about import alerts affecting certain Olympus medical devices manufactured in Japan. The FDA said the alerts were issued because of ongoing concerns about outstanding Quality System regulation violations by Olympus and its subsidiaries.
The affected product categories included certain bronchoscopes, ureterorenoscopes, laparoscopes and automated endoscope reprocessors. Olympus acknowledged that the FDA action prevented the importation of specified devices into the United States until further notice.
These FDA warnings may help show that infection risks involving Olympus devices were not isolated concerns. For patients who developed infections after endoscopic procedures, medical records, procedure reports and infection timelines may help determine whether an Olympus device or accessory was involved.
The following records may be important when evaluating a potential claim:
Because patients may not know which device was used, the first step is often to determine whether the procedure involved an Olympus scope or an accessory.
Patients trust medical devices to be safe. When a device used during a routine procedure exposes someone to dangerous bacteria, the consequences can be devastating. Our law firm has significant experience handling these types of claims and helping thousands of patients recover billions of dollars. Schedule a free consultation with a member of Olympus Scopes Lawsuit team. Call 800-359-5690 for legal representation with no out-of-pocket costs.
The Carlson Law Firm is reviewing claims involving serious infections, sepsis and deaths after procedures involving Olympus scopes or Olympus endoscope accessories. If you or a loved one suffered a serious infection after a medical procedure involving a scope, your records may help determine whether an Olympus device was involved.
We care. We can help.